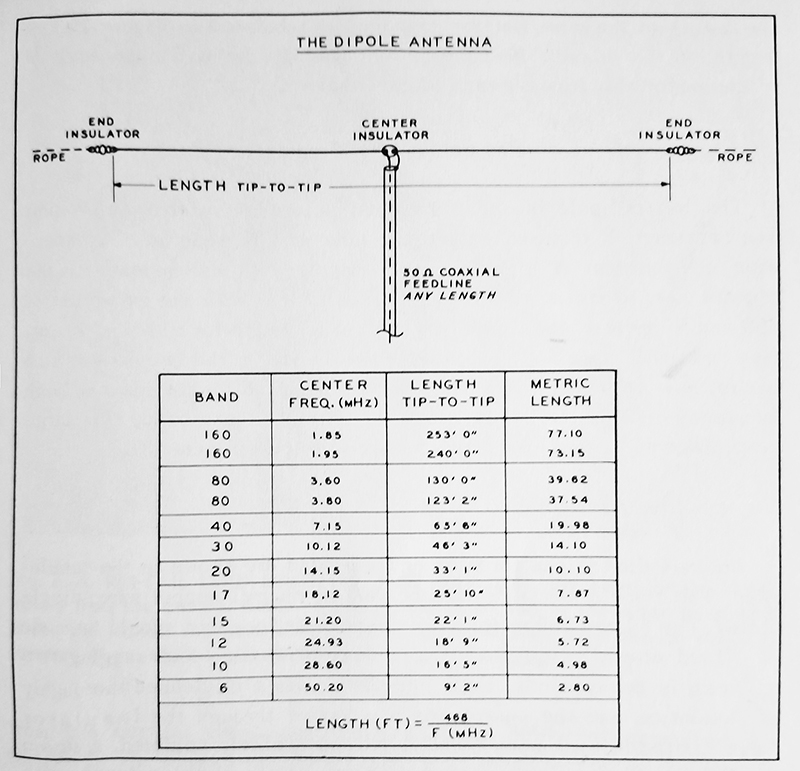
Dipole Length Chart
Dipole Length Chart - → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→ r 2 r3 2 given q and d, if you know. Which intermolecular forces in h2o make ice less dense than liquid water: Two charges alone (one positive and one negative) will not be stable. When the length of carbon chain increases the van der waals dispersion forces increase which results in an increase in the strength and size of the temporarily induced dipole. Solvents that contain a strong dipole moment but don't participate in hydrogen bonding (ie. Is ch3co2h a dipole dipole? It’s net dipole moment is not zero. What type of intermolecular force would water. For an electric dipole to exist an external electric field is required. Van der waals forces are considered to be intermolecular forces, along with hydrogen bonds dipole dipole forces, these forces tend to bond or attract molecules to molecules. Solvents that contain a strong dipole moment but don't participate in hydrogen bonding (ie. When the length of carbon chain increases the van der waals dispersion forces increase which results in an increase in the strength and size of the temporarily induced dipole. Dipoles are created when one element has a higher electronegativity (tendency to attract electrons) than another in a bond. For an electric dipole to exist an external electric field is required. It’s net dipole moment is not zero. Two charges alone (one positive and one negative) will not be stable. The dipole moment of pcl_3 is greater. These dipoles may be permanent (polar molecules) or. This is because the charges will. > bb (ph_3) the electron geometry of ph_3 is tetrahedral and the molecular geometry is trigonal pyramidal. The dipole moment of pcl_3 is greater. → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→ r 2 r3 2 given q and d, if you know. What type of intermolecular force would water. Donate or accept protons with itself) dichloromethane and acetone are good. When the length of. The electronegativity difference has to. Is ch3co2h a dipole dipole? Which intermolecular forces in h2o make ice less dense than liquid water: When the length of carbon chain increases the van der waals dispersion forces increase which results in an increase in the strength and size of the temporarily induced dipole. Donate or accept protons with itself) dichloromethane and acetone. These dipoles may be permanent (polar molecules) or. Solvents that contain a strong dipole moment but don't participate in hydrogen bonding (ie. Which intermolecular forces in h2o make ice less dense than liquid water: This is because the charges will. The dipole moment of pcl_3 is greater. > bb (ph_3) the electron geometry of ph_3 is tetrahedral and the molecular geometry is trigonal pyramidal. When the length of carbon chain increases the van der waals dispersion forces increase which results in an increase in the strength and size of the temporarily induced dipole. Is ch3co2h a dipole dipole? → e = → e 1 + → e. What type of intermolecular force would water. The dipole field at a point located at → r away from the origin set at the center of a dipole is: Donate or accept protons with itself) dichloromethane and acetone are good. → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→. This is because the charges will. What type of intermolecular force would water. > bb (ph_3) the electron geometry of ph_3 is tetrahedral and the molecular geometry is trigonal pyramidal. The dipole field at a point located at → r away from the origin set at the center of a dipole is: Two charges alone (one positive and one negative). → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→ r 2 r3 2 given q and d, if you know. These dipoles may be permanent (polar molecules) or. Solvents that contain a strong dipole moment but don't participate in hydrogen bonding (ie. Van der waals forces are considered to. The dipole field at a point located at → r away from the origin set at the center of a dipole is: Which intermolecular forces in h2o make ice less dense than liquid water: The dipole moment of pcl_3 is greater. Two charges alone (one positive and one negative) will not be stable. It’s net dipole moment is not zero. → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→ r 2 r3 2 given q and d, if you know. The dipole moment of pcl_3 is greater. The electronegativity difference has to. This is because the charges will. It’s net dipole moment is not zero. Van der waals forces are considered to be intermolecular forces, along with hydrogen bonds dipole dipole forces, these forces tend to bond or attract molecules to molecules. Which intermolecular forces in h2o make ice less dense than liquid water: → e = → e 1 + → e 2 → e = kq→ r 1 r3 1 − kq→ r 2 r3 2 given q and d, if you know. This is because the charges will. Is ch3co2h a dipole dipole? The electronegativity difference has to. The dipole field at a point located at → r away from the origin set at the center of a dipole is: Dipoles are created when one element has a higher electronegativity (tendency to attract electrons) than another in a bond. Two charges alone (one positive and one negative) will not be stable. The dipole moment of pcl_3 is greater. Donate or accept protons with itself) dichloromethane and acetone are good. > bb (ph_3) the electron geometry of ph_3 is tetrahedral and the molecular geometry is trigonal pyramidal. It’s net dipole moment is not zero. Solvents that contain a strong dipole moment but don't participate in hydrogen bonding (ie.Dipole Antenna Length Chart
Dipole Antenna Length Calculator
Dipole Length Chart Ponasa
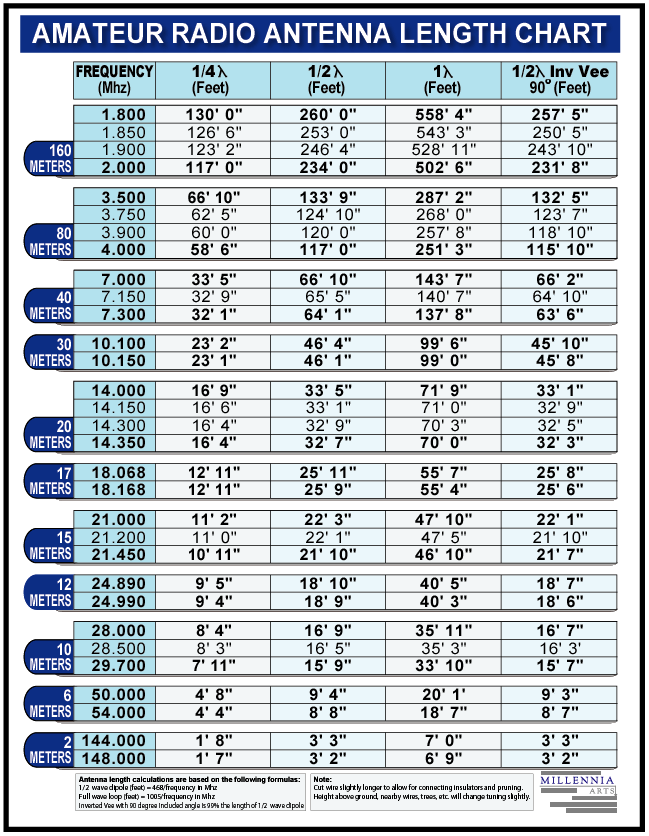
VHF Dipole Antenna Length Calculator
Dipole Length Chart Portal.posgradount.edu.pe
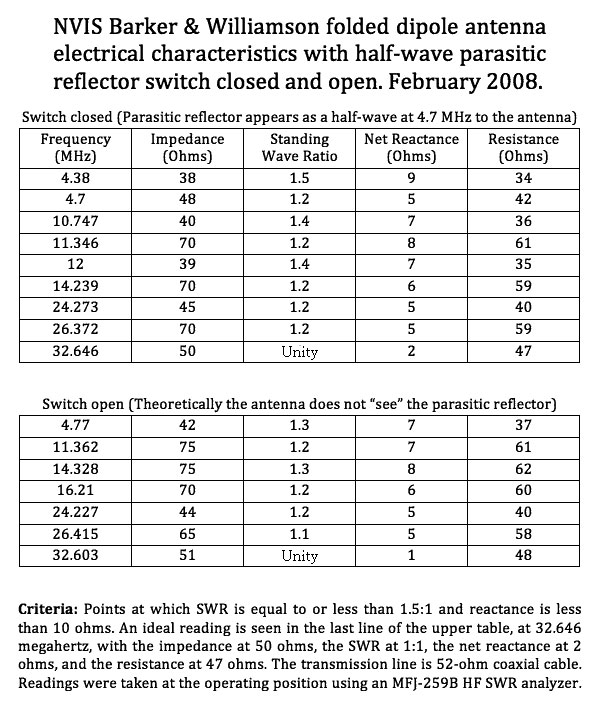
dipole antenna length chart Coaxial folded dipole antenna
Dipole Length Chart Ponasa
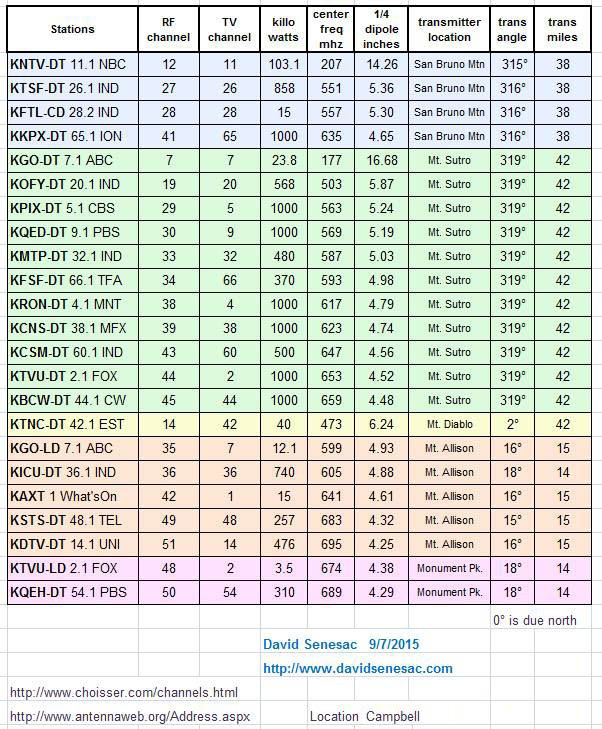
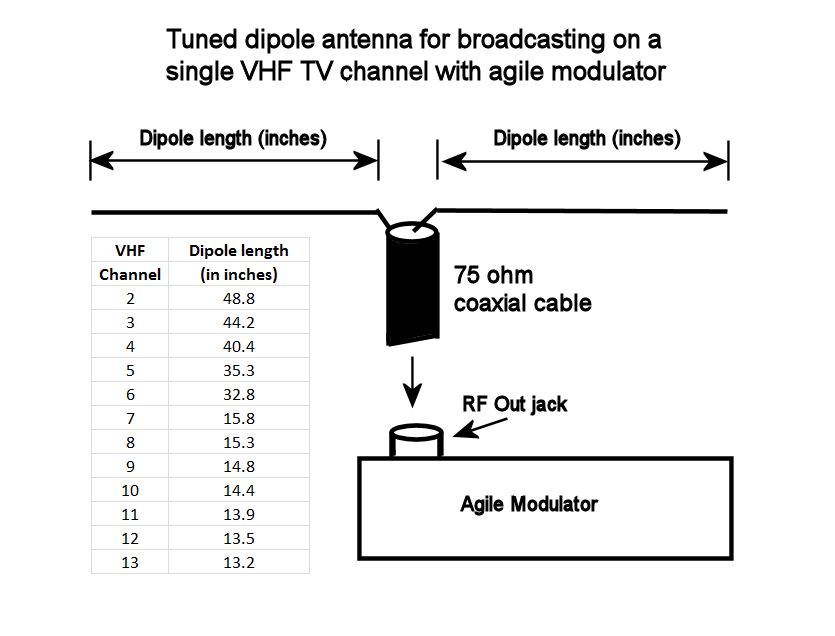
Creating a Home TV Transmitter
Comparative chart for straight and Vdipole antennas. Download Table
For An Electric Dipole To Exist An External Electric Field Is Required.
These Dipoles May Be Permanent (Polar Molecules) Or.
When The Length Of Carbon Chain Increases The Van Der Waals Dispersion Forces Increase Which Results In An Increase In The Strength And Size Of The Temporarily Induced Dipole.
What Type Of Intermolecular Force Would Water.
Related Post: