Electronegativity Trend Chart
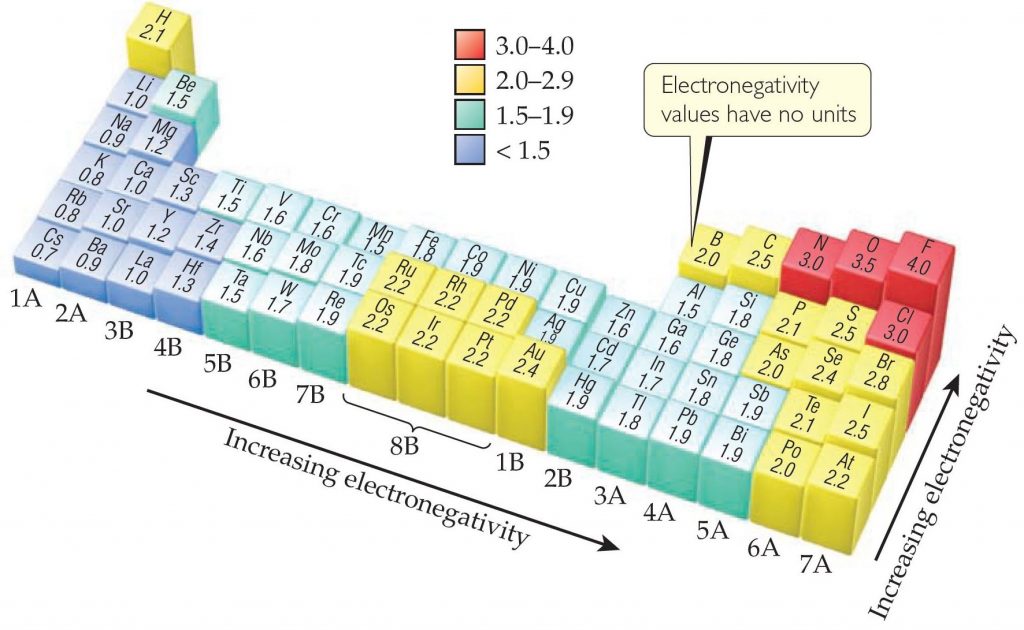
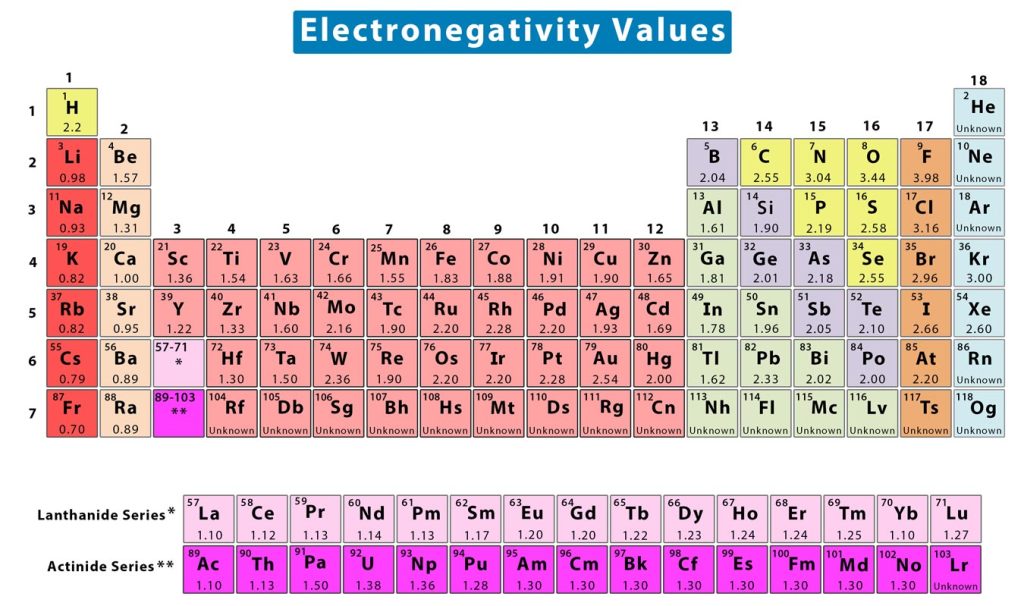
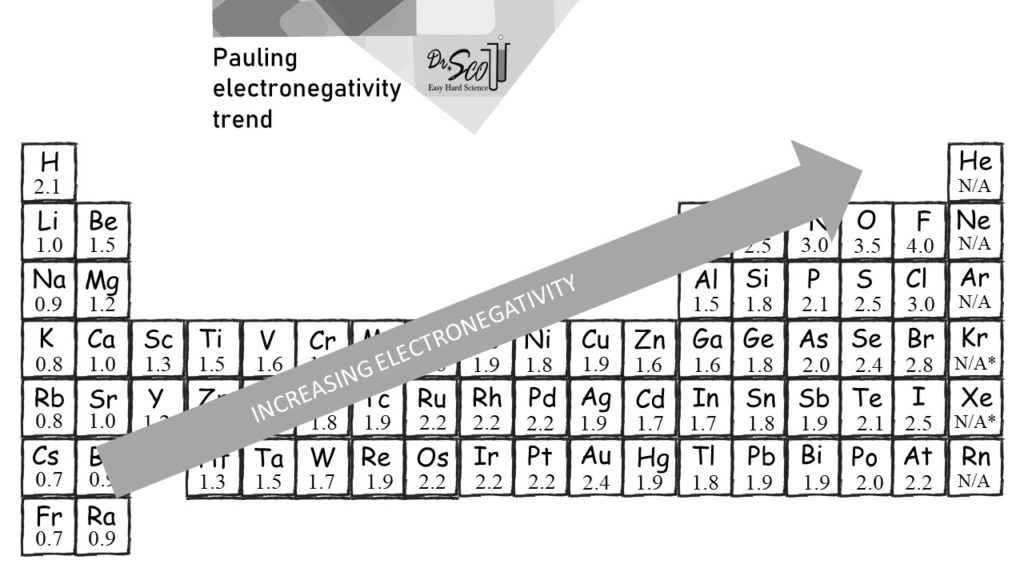
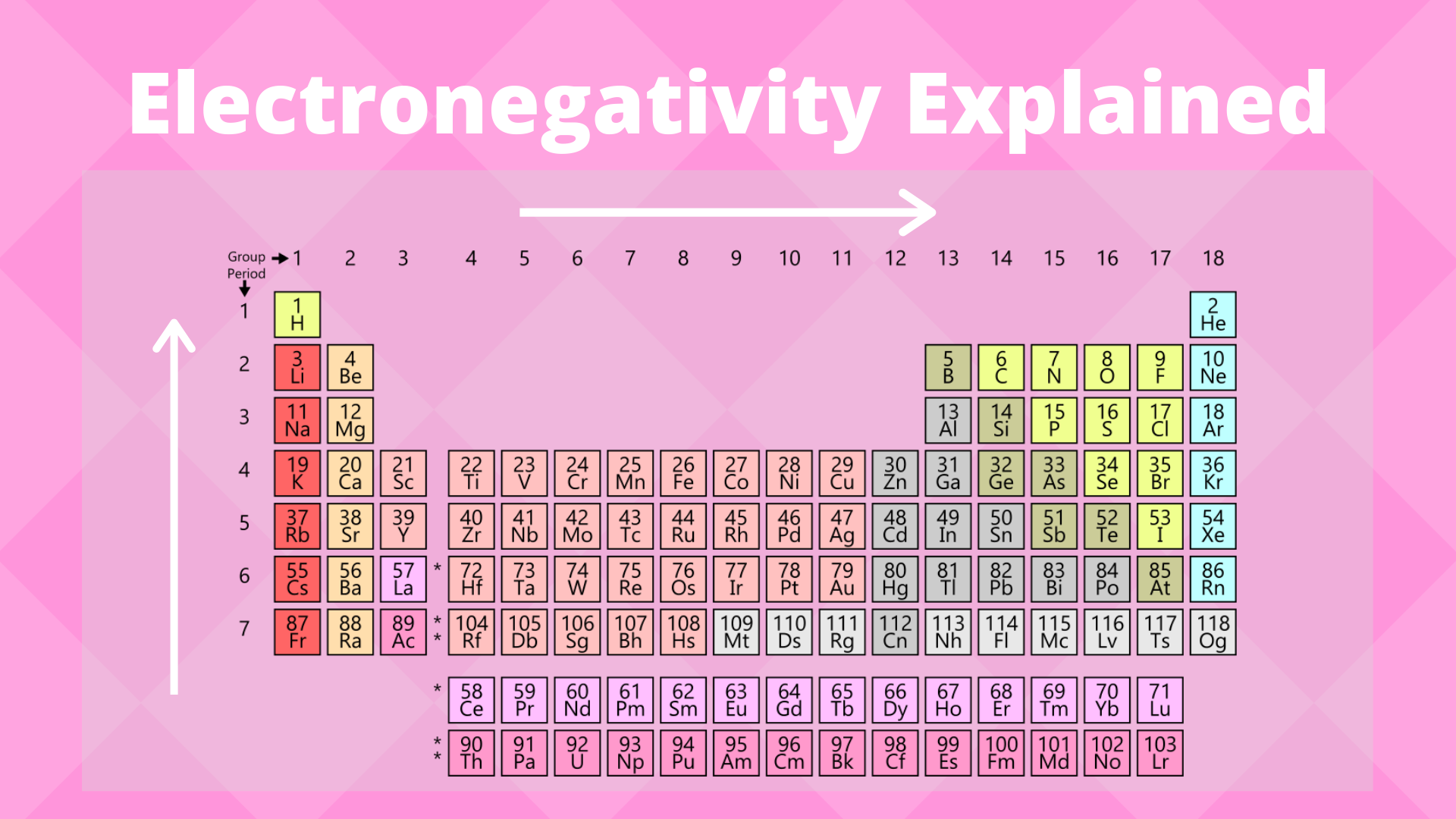
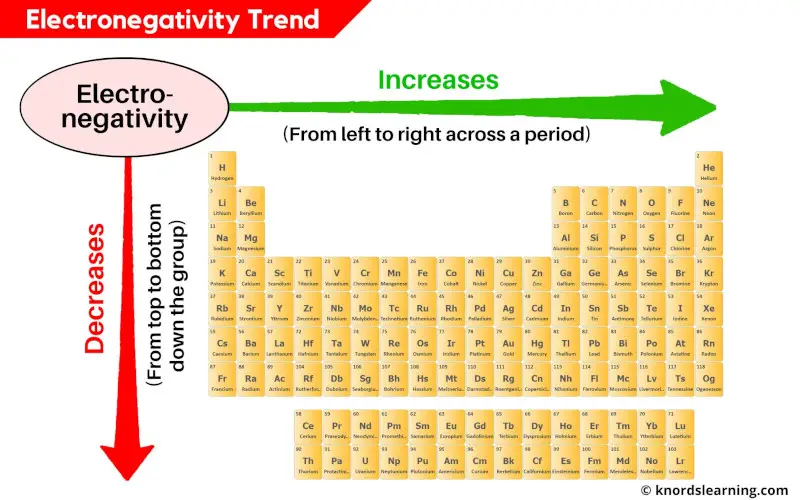
Electronegativity Trend Chart - In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. The electronegativity difference serves as a measure of percentage at which a bond is ionic.roughly speaking, electro negativity difference of 1.7 is equivalent to 50 ℅ ionic. Covalent bonds, and discover how to predict. I guess this electronegativity comes from a few aspects. See an electronegativity example, and discover how to find electronegativity using the right tools for. Learn what ionic character is. Noble gases are supposed to be happy with the amount of electrons they have, because they have 8 valence electrons (thus, most noble. Why do krypton and xenon have high electronegativity? 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds. See an electronegativity example, and discover how to find electronegativity using the right tools for. I guess this electronegativity comes from a few aspects. Learn what ionic character is. Covalent bonds, and discover how to predict. The electronegativity difference serves as a measure of percentage at which a bond is ionic.roughly speaking, electro negativity difference of 1.7 is equivalent to 50 ℅ ionic. In this sense, elements are less electronegative (or more electropositive) as you go down any group in the periodic table. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. Electronegativity increases across a period However, the difference in electronegativity between. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. Learn what ionic character is. See an electronegativity example, and discover how to find electronegativity using the right tools for. In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds.. If we take the most simple definition, that it is the sum of the electron affinity and the ionization energy (divided by two), we. Covalent bonds, and discover how to predict. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. However, the difference in electronegativity between. Why do krypton and xenon. However, the difference in electronegativity between. I guess this electronegativity comes from a few aspects. Trace ionic character trend on the periodic table, examine ionic vs. 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. See an electronegativity. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. In this sense, elements are less electronegative (or more electropositive) as you go down any group in the periodic table. I guess this electronegativity comes from a few aspects. The electronegativity difference serves as a measure of percentage at which a bond. Learn what ionic character is. The electronegativity difference serves as a measure of percentage at which a bond is ionic.roughly speaking, electro negativity difference of 1.7 is equivalent to 50 ℅ ionic. In this sense, elements are less electronegative (or more electropositive) as you go down any group in the periodic table. If we take the most simple definition, that. In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds. 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. Why do krypton and xenon have high. Electronegativity increases across a period The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. If we take the most simple. Covalent bonds, and discover how to predict. If we take the most simple definition, that it is the sum of the electron affinity and the ionization energy (divided by two), we. See an electronegativity example, and discover how to find electronegativity using the right tools for. Trace ionic character trend on the periodic table, examine ionic vs. Noble gases are. The trend in electronegativity can be seen on the periodic table and, more specifically, in the following graphs. 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. Noble gases are supposed to be happy with the amount of. In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds. However, the difference in electronegativity between. Learn what ionic character is. See an electronegativity example, and discover how to find electronegativity using the right tools for. Why do krypton and xenon have high electronegativity? If we take the most simple definition, that it is the sum of the electron affinity and the ionization energy (divided by two), we. I guess this electronegativity comes from a few aspects. Electronegativity increases across a period In the 1930s, scientist linus pauling proposed a scale to measure and explain the attraction atoms have for valence electrons in bonds. In this sense, elements are less electronegative (or more electropositive) as you go down any group in the periodic table. The electronegativity difference serves as a measure of percentage at which a bond is ionic.roughly speaking, electro negativity difference of 1.7 is equivalent to 50 ℅ ionic. Learn what ionic character is. Covalent bonds, and discover how to predict. Noble gases are supposed to be happy with the amount of electrons they have, because they have 8 valence electrons (thus, most noble. 1 following is from wikipedia electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical. Trace ionic character trend on the periodic table, examine ionic vs. Why do krypton and xenon have high electronegativity?Electronegativity Definition, Value Chart, and Trend in Periodic Table
Periodic Table Electronegativity Printable
Electronegativity Chart Printable
Electronegativity Definition, Value Chart, and Trend in Periodic Table
Periodic Table Including Electronegativity
Periodic Trends in Electronegativity CK12 Foundation
Electronegativity Trend Science Trends
All Periodic Trends of Periodic Table (Simple Explanation)
Electronegativity Definition and Trend Periodic table, Important life lessons, Periodic table
Periodic Table of Electronegativities
However, The Difference In Electronegativity Between.
The Trend In Electronegativity Can Be Seen On The Periodic Table And, More Specifically, In The Following Graphs.
See An Electronegativity Example, And Discover How To Find Electronegativity Using The Right Tools For.
Related Post:




.PNG)