Electronic And Molecular Geometry Chart
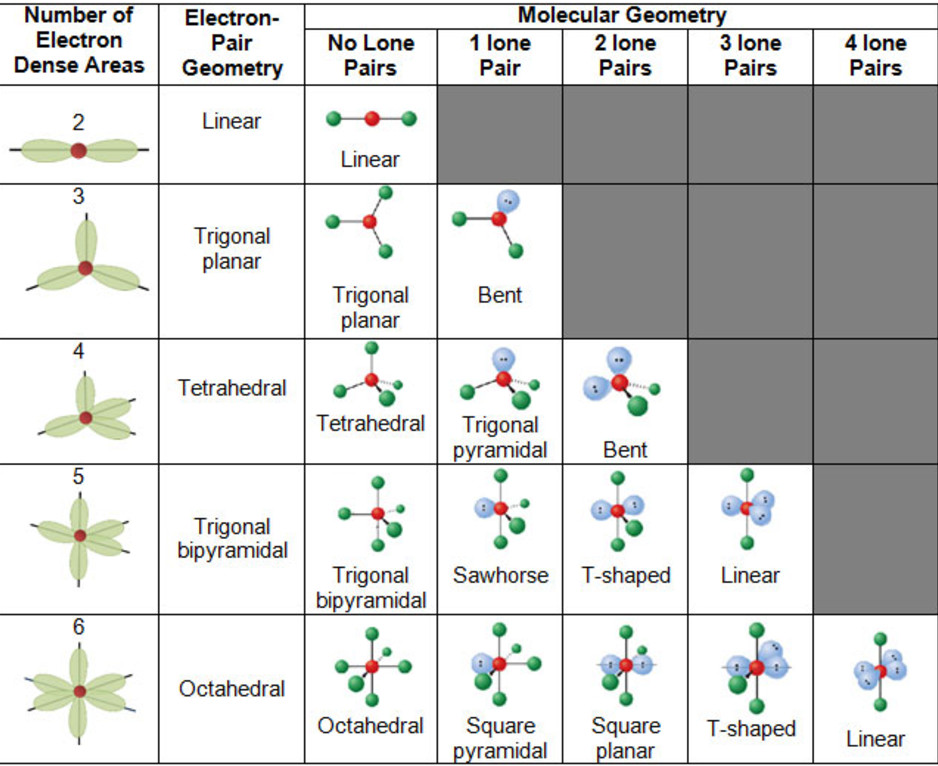
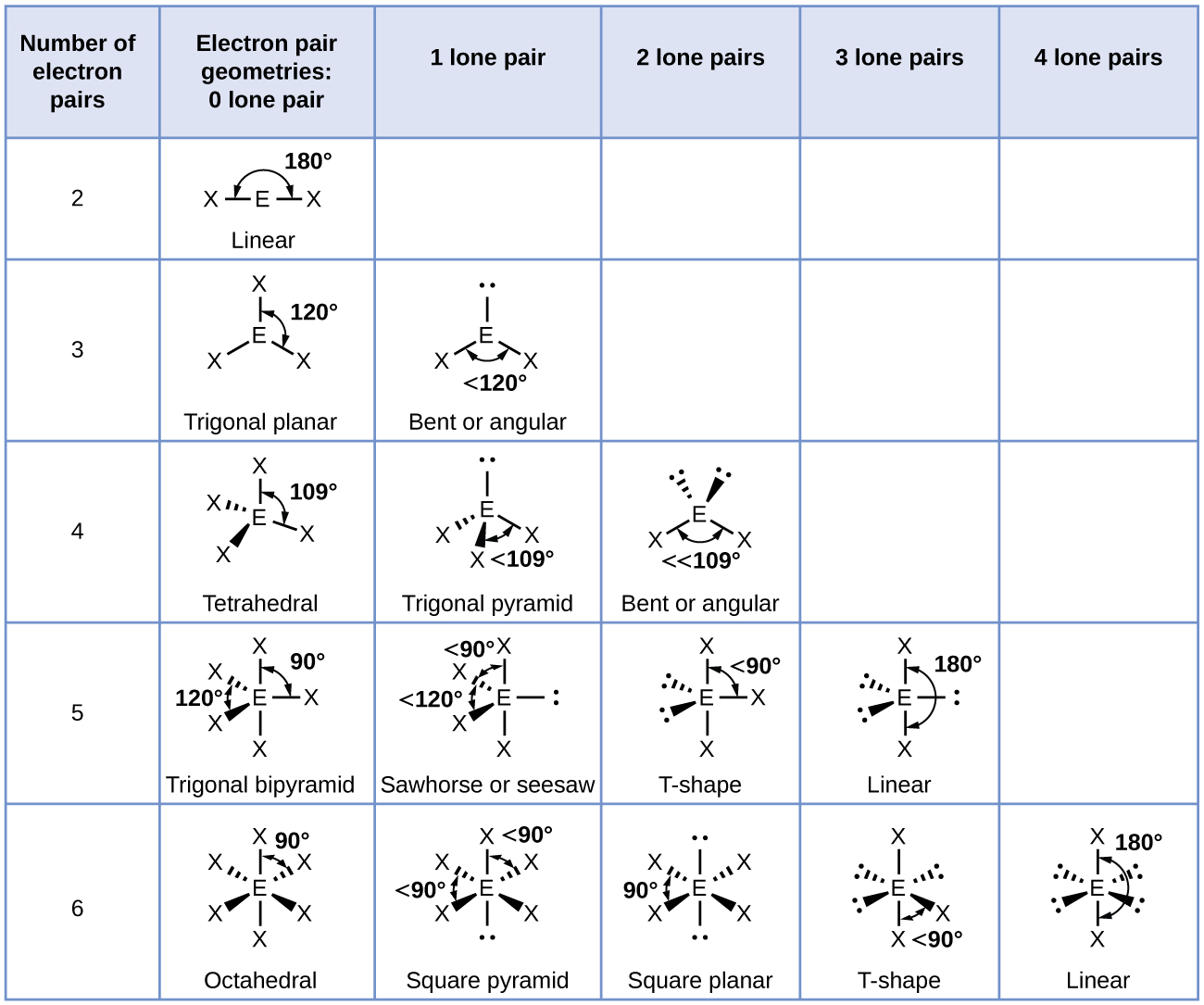
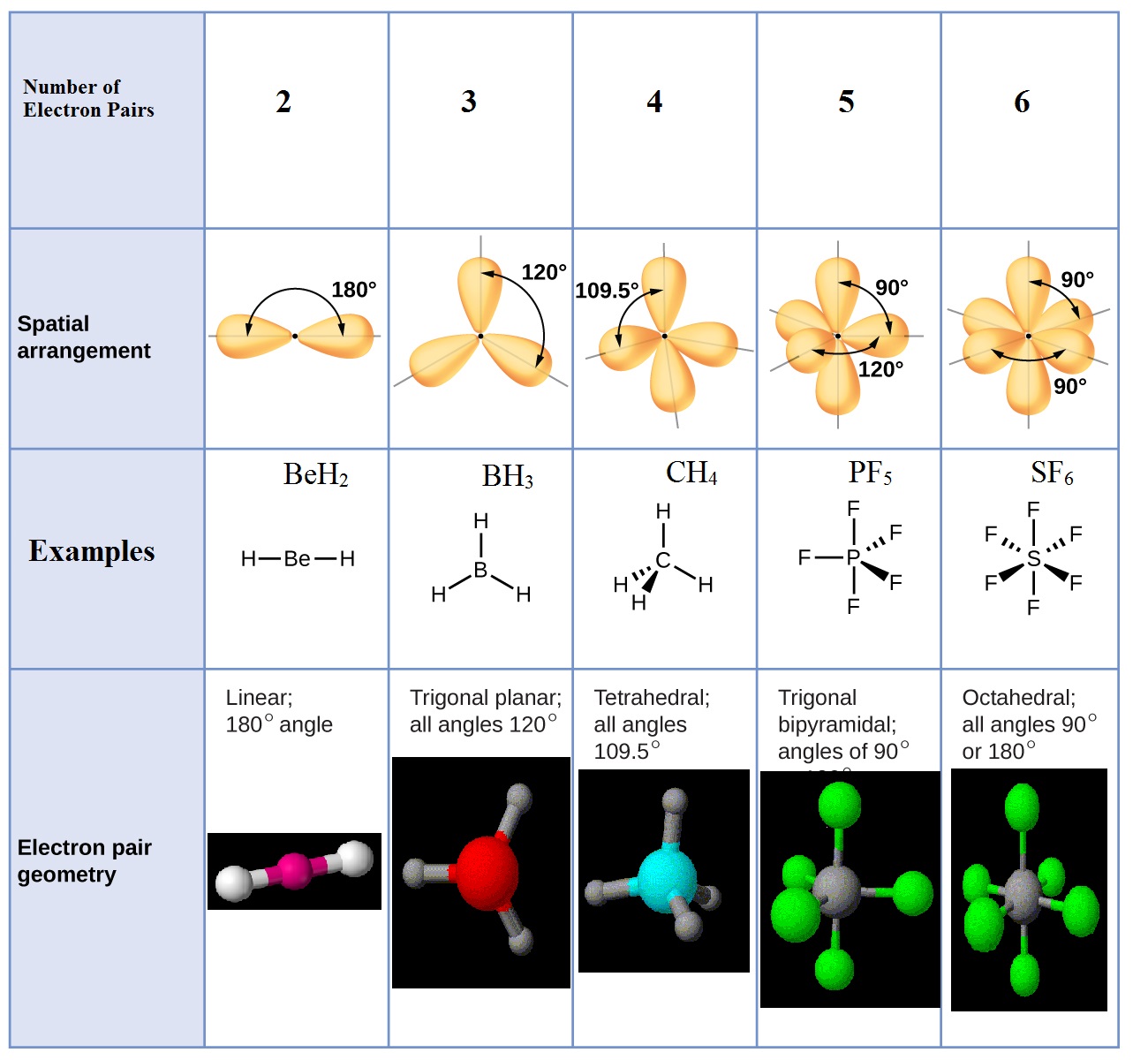
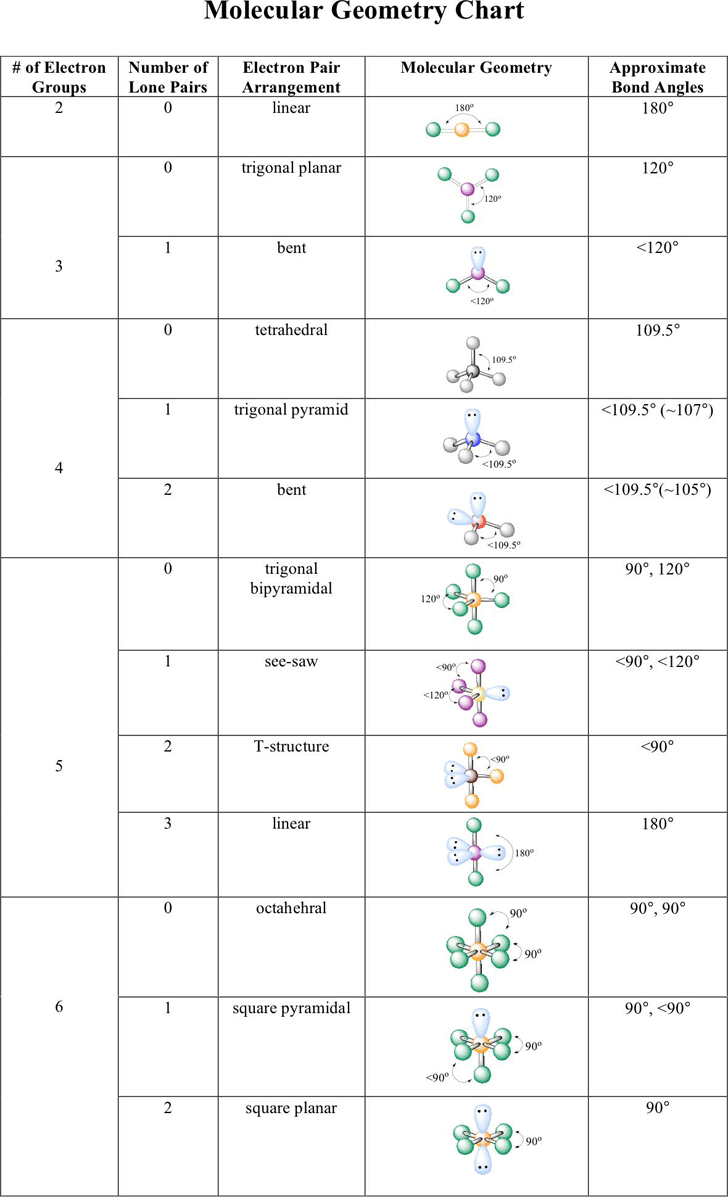
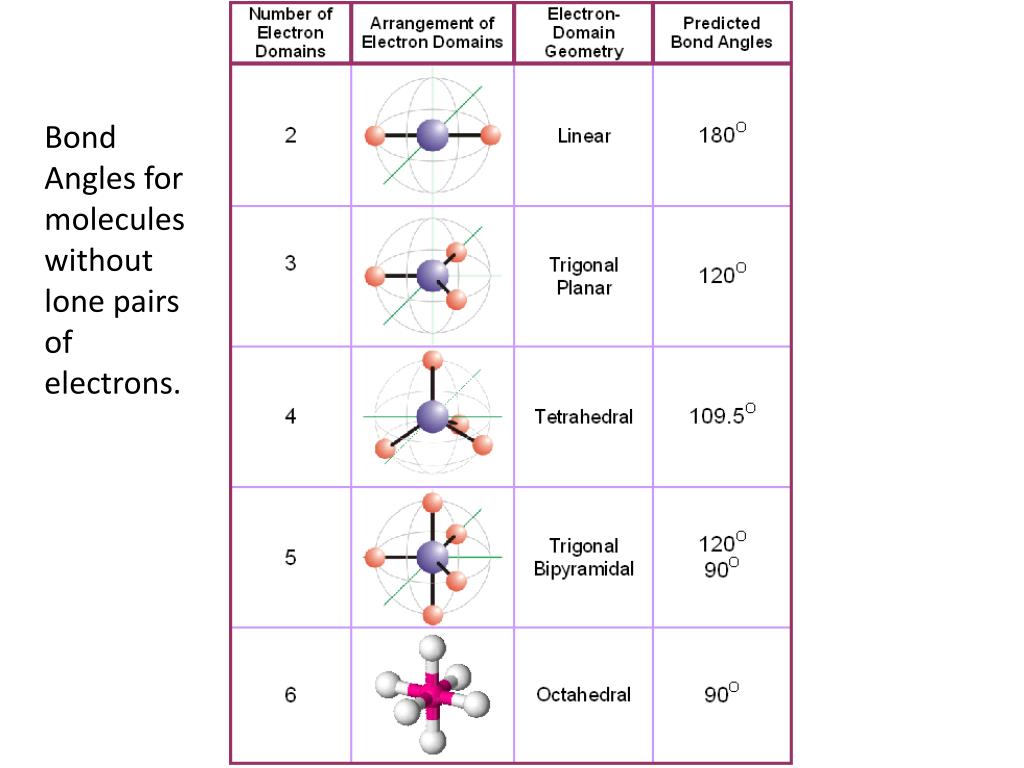
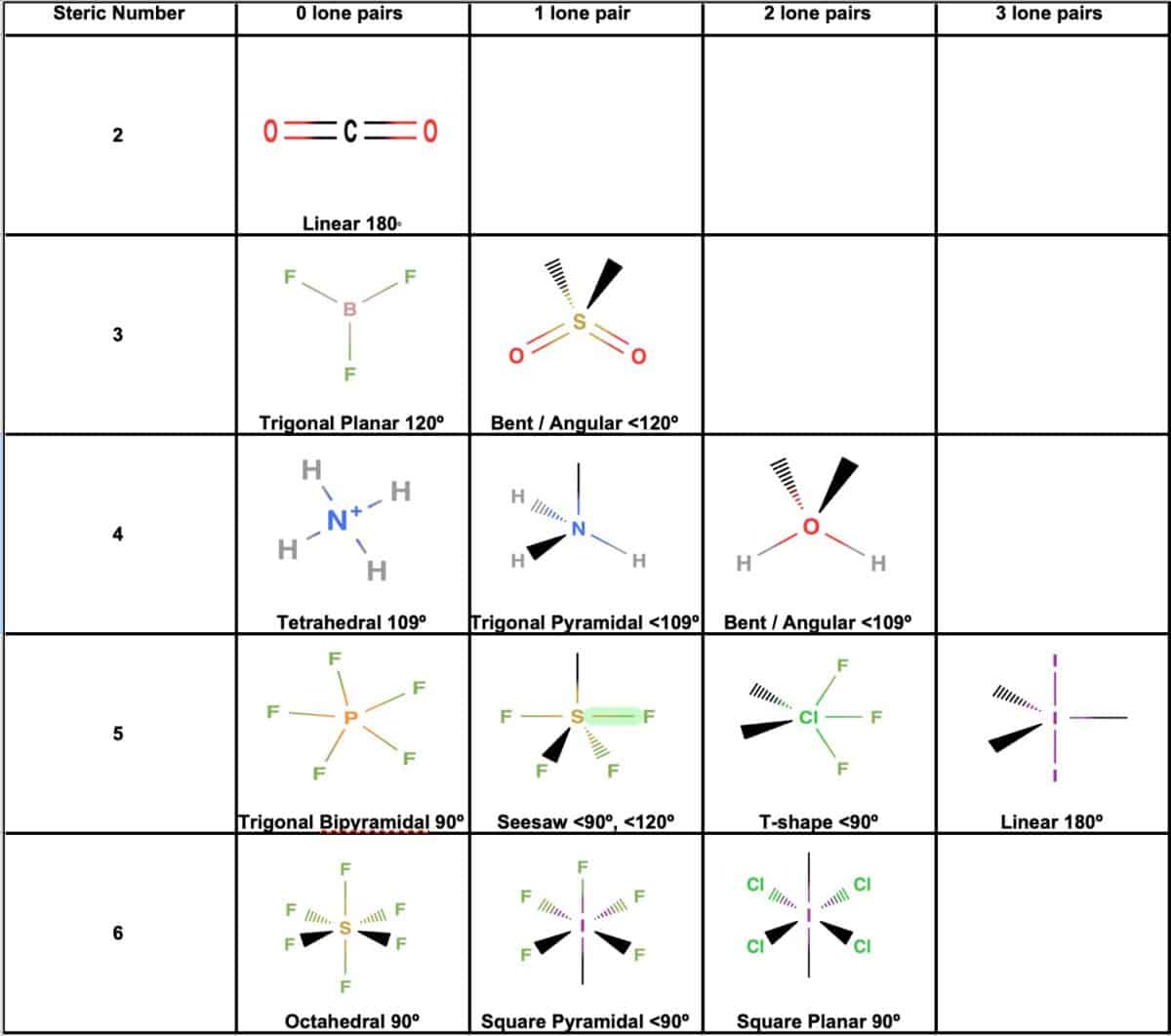
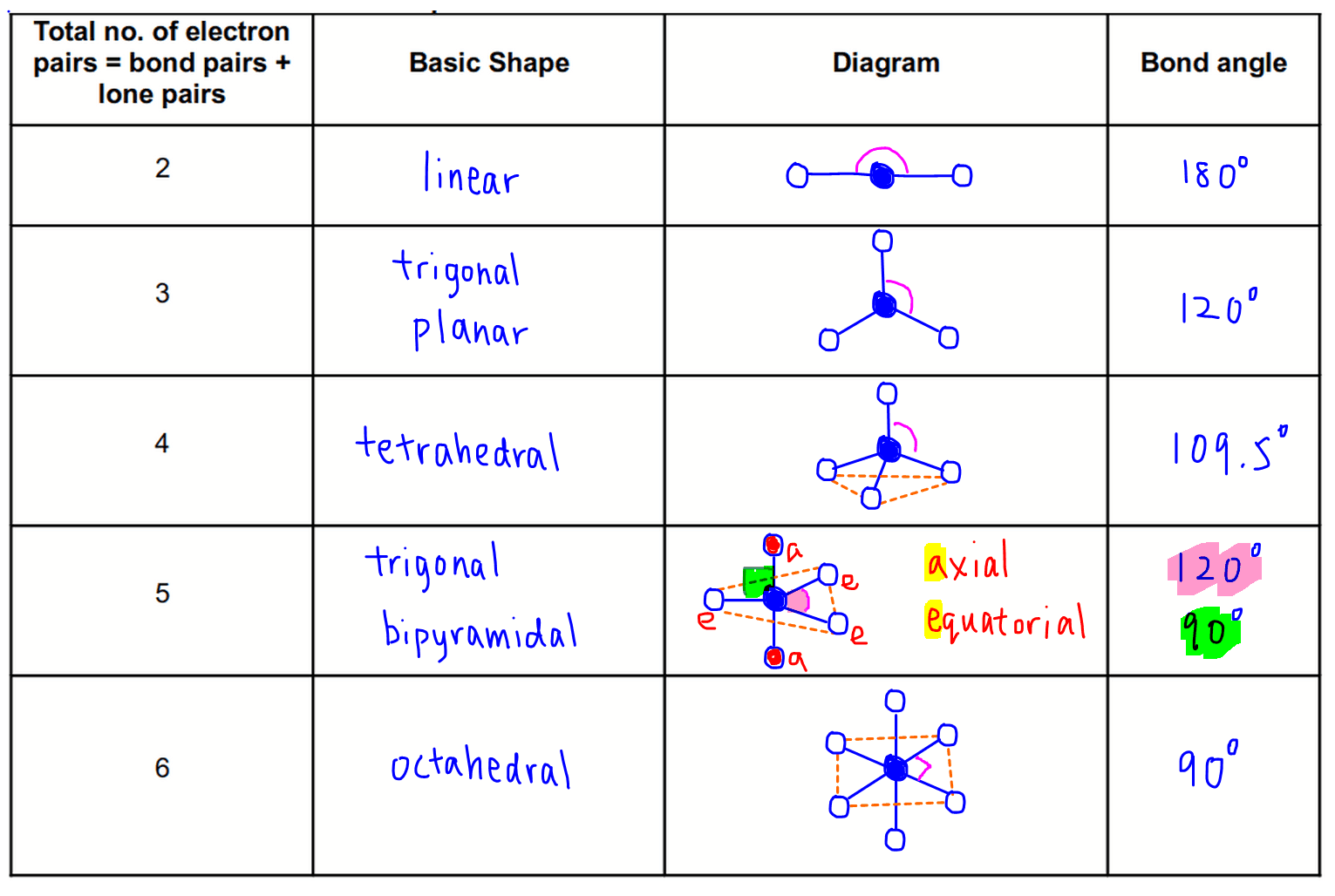
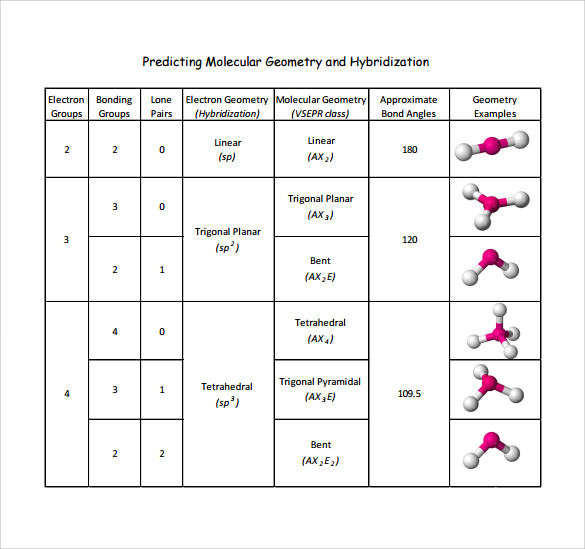
Electronic And Molecular Geometry Chart - Molecular geometry, on the other hand, helps us in determining the entire atom and its configuration. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Abnem a = central atom, b = directly bonded atoms to a, and e = nonbonding (unshared) pairs of electrons *note that a molecule formed by joining only two (2). What are the different shapes of molecules. Determine the electron geometry from the lewis dot structure. Students and scientists can use these charts to create three. Check out a table of molecular geometries with examples and diagrams. In chemistry, vsepr theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing. Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes). A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. You can manipulate the number of single, double, or triple bonds and lone. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. Students and scientists can use these charts to create three. In chemistry, vsepr theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing. A group of electrons is. Electron geometry helps us in determining the arrangement of various electron groups. Determine the electron geometry from the lewis dot structure. There is a three step approach to determining the geometry of a molecule. Molecular geometry, on the other hand, helps us in determining the entire atom and its configuration. What are the different shapes of molecules. Abnem a = central atom, b = directly bonded atoms to a, and e = nonbonding (unshared) pairs of electrons *note that a molecule formed by joining only two (2). Electron geometry helps us in determining the arrangement of various electron groups. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Here we will discuss. Check out a table of molecular geometries with examples and diagrams. What are the different shapes of molecules. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. You can manipulate the number of single, double, or triple bonds and lone. A molecular geometry chart is a collection of rules on how molecules and electrons will. Determine the electron geometry from the lewis dot structure. There is a three step approach to determining the geometry of a molecule. Abnem a = central atom, b = directly bonded atoms to a, and e = nonbonding (unshared) pairs of electrons *note that a molecule formed by joining only two (2). Molecular geometry, on the other hand, helps us. You can manipulate the number of single, double, or triple bonds and lone. What are the different shapes of molecules. Abnem a = central atom, b = directly bonded atoms to a, and e = nonbonding (unshared) pairs of electrons *note that a molecule formed by joining only two (2). A group of electrons is. Check out a table of. Students and scientists can use these charts to create three. Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes). Molecular geometry, on the other hand, helps us in determining the entire atom and its configuration. In chemistry, vsepr theory is based on the principle that each. Check out a table of molecular geometries with examples and diagrams. Electron geometry helps us in determining the arrangement of various electron groups. Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes). In chemistry, vsepr theory is based on the principle that each atom in a. Students and scientists can use these charts to create three. There is a three step approach to determining the geometry of a molecule. You can manipulate the number of single, double, or triple bonds and lone. Determine the electron geometry from the lewis dot structure. A group of electrons is. Check out a table of molecular geometries with examples and diagrams. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes). There is a three step approach. What are the different shapes of molecules. Students and scientists can use these charts to create three. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. Molecular geometry, on the other hand, helps us in determining the entire atom and its configuration. Determine the electron geometry from the lewis. Students and scientists can use these charts to create three. In chemistry, vsepr theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing. What are the different shapes of molecules. Here we will discuss both electron pair geometries (also called electron group or electron. Explore our table of common electron geometries with bonding domains, bond angles, and formulas. Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes). There is a three step approach to determining the geometry of a molecule. Abnem a = central atom, b = directly bonded atoms to a, and e = nonbonding (unshared) pairs of electrons *note that a molecule formed by joining only two (2). Check out a table of molecular geometries with examples and diagrams. Determine the electron geometry from the lewis dot structure. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. Electron geometry helps us in determining the arrangement of various electron groups. Molecular geometry, on the other hand, helps us in determining the entire atom and its configuration. Determine the lewis dot structure of the compound. In chemistry, vsepr theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing. A group of electrons is.Valence Shell Electron Pair Repulsion Theory Chemical Bonding and Molecular Structure
Electron and molecular geometry chart examples michaelhost
Electron and Molecular Geometries … Molecular geometry, Teaching chemistry, Chemistry lessons
Electron pair geometry and molecular geometry chart volfplus
Molecular Geometry Chart and linear molecules
Molecular Geometry Chart Template Free Download Speedy Template
Electron and molecular geometry chart examples heryhd
Molecular Geometry Chart and linear molecules
Electron vs molecular geometry chart stopfiko
FREE 8+ Sample Molecular Geometry Chart Templates in PDF MS Word
What Are The Different Shapes Of Molecules.
You Can Manipulate The Number Of Single, Double, Or Triple Bonds And Lone.
Students And Scientists Can Use These Charts To Create Three.
Related Post: