Nt8 Live Performance Chart
Nt8 Live Performance Chart - From these three ingredients, we get elemental chlorine (cl 2), caustic (often sodium hydroxide or naoh). Thirty five million tons of chlorine were prepared by. It is the technology used to produce chlorine and sodium hydroxide (caustic soda), [1] which are commodity chemicals required by industry. Sodium chloride (nacl) is the primary salt used; Released as a gas at the anode. Large amounts of caustic soda. Electrolysis of salt brines produces chlorine and caustic. The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main electrolytic processes: It is produced in liquid form and is. H _ { 2 } gas is obtained at cathode, chlorine gas at. Generated at the cathode as a valuable byproduct. Thirty five million tons of chlorine were prepared by. Sodium chloride (nacl) is the primary salt used; The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main electrolytic processes: Chlorine gas is obtained at anode as a byproduct. From these three ingredients, we get elemental chlorine (cl 2), caustic (often sodium hydroxide or naoh). It is produced in liquid form and is. In my limited chemical world, i came across a unique relationship between caustic soda & chlorine. H _ { 2 } gas is obtained at cathode, chlorine gas at. There are three key ingredients to make elemental chlorine; H _ { 2 } gas is obtained at cathode, chlorine gas at. Has developed a catalytic process that is highly recognized and applied worldwide as a technology for recycling the hydrogen chloride generated as by. Released as a gas at the anode. Also known as the chloralkali industry, in the process of manufacturing caustic. Large amounts of caustic soda. In my limited chemical world, i came across a unique relationship between caustic soda & chlorine. Thirty five million tons of chlorine were prepared by. It is produced in liquid form and is. Also known as the chloralkali industry, in the process of manufacturing caustic. The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main. Chlorine gas is obtained at anode as a byproduct. Sodium chloride (nacl) is the primary salt used; From these three ingredients, we get elemental chlorine (cl 2), caustic (often sodium hydroxide or naoh). Large amounts of caustic soda. In all 3 methods, the chlorine (cl2) is produced at the positive electrode (anode) and the caustic soda (naoh) and hydrogen (h2). It yields caustic soda (naoh). There are three key ingredients to make elemental chlorine; Chlorine gas is obtained at anode as a byproduct. H _ { 2 } gas is obtained at cathode, chlorine gas at. Sodium chloride (nacl) is the primary salt used; Electrolysis of salt brines produces chlorine and caustic. Generated at the cathode as a valuable byproduct. Released as a gas at the anode. It yields caustic soda (naoh). From these three ingredients, we get elemental chlorine (cl 2), caustic (often sodium hydroxide or naoh). Large amounts of caustic soda. In all 3 methods, the chlorine (cl2) is produced at the positive electrode (anode) and the caustic soda (naoh) and hydrogen (h2) are produced, directly or indirectly, at the negative electrode. It is the technology used to produce chlorine and sodium hydroxide (caustic soda), [1] which are commodity chemicals required by industry. Thirty five million. Also known as the chloralkali industry, in the process of manufacturing caustic. The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main electrolytic processes: There are three key ingredients to make elemental chlorine; Released as a gas at the anode. Chlorine gas is obtained at anode as a byproduct. However, the use of potassium chloride. Generated at the cathode as a valuable byproduct. Also known as the chloralkali industry, in the process of manufacturing caustic. Sodium chloride (nacl) is the primary salt used; Chlorine gas is obtained at anode as a byproduct. The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main electrolytic processes: H _ { 2 } gas is obtained at cathode, chlorine gas at. Thirty five million tons of chlorine were prepared by. Released as a gas at the anode. Generated at the cathode as a valuable byproduct. In all 3 methods, the chlorine (cl2) is produced at the positive electrode (anode) and the caustic soda (naoh) and hydrogen (h2) are produced, directly or indirectly, at the negative electrode. It yields caustic soda (naoh). Generated at the cathode as a valuable byproduct. Chlorine gas is obtained at anode as a byproduct. In my limited chemical world, i came. Released as a gas at the anode. It is produced in liquid form and is. Sodium chloride (nacl) is the primary salt used; It yields caustic soda (naoh). There are three key ingredients to make elemental chlorine; In my limited chemical world, i came across a unique relationship between caustic soda & chlorine. Electrolysis of salt brines produces chlorine and caustic. In all 3 methods, the chlorine (cl2) is produced at the positive electrode (anode) and the caustic soda (naoh) and hydrogen (h2) are produced, directly or indirectly, at the negative electrode. From these three ingredients, we get elemental chlorine (cl 2), caustic (often sodium hydroxide or naoh). The document summarizes the production of chlorine and sodium hydroxide (caustic soda) via three main electrolytic processes: Chlorine gas is obtained at anode as a byproduct. Also known as the chloralkali industry, in the process of manufacturing caustic. Has developed a catalytic process that is highly recognized and applied worldwide as a technology for recycling the hydrogen chloride generated as by. Large amounts of caustic soda. However, the use of potassium chloride.How to Add Indicators on Chart NT8 YouTube
Chart Tools (NT8) Chart Dynamix
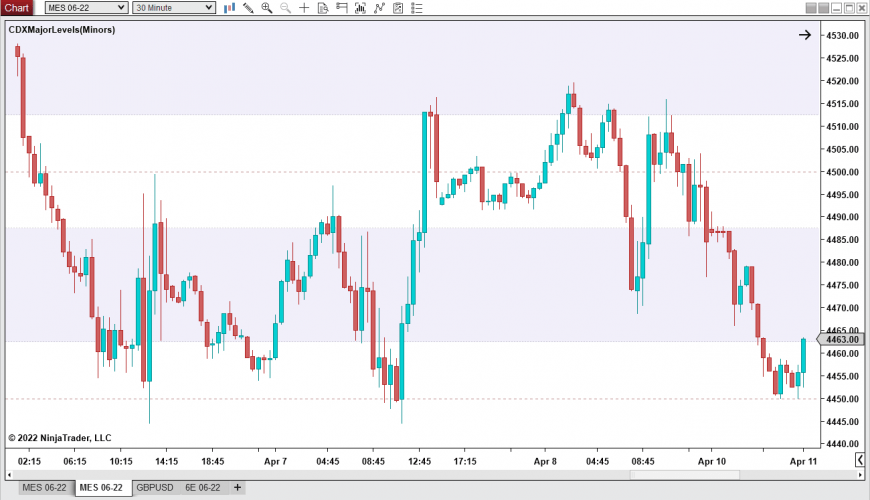
Major Levels (NT8) Chart Dynamix
WaveTrend™ (NT8) Chart Dynamix
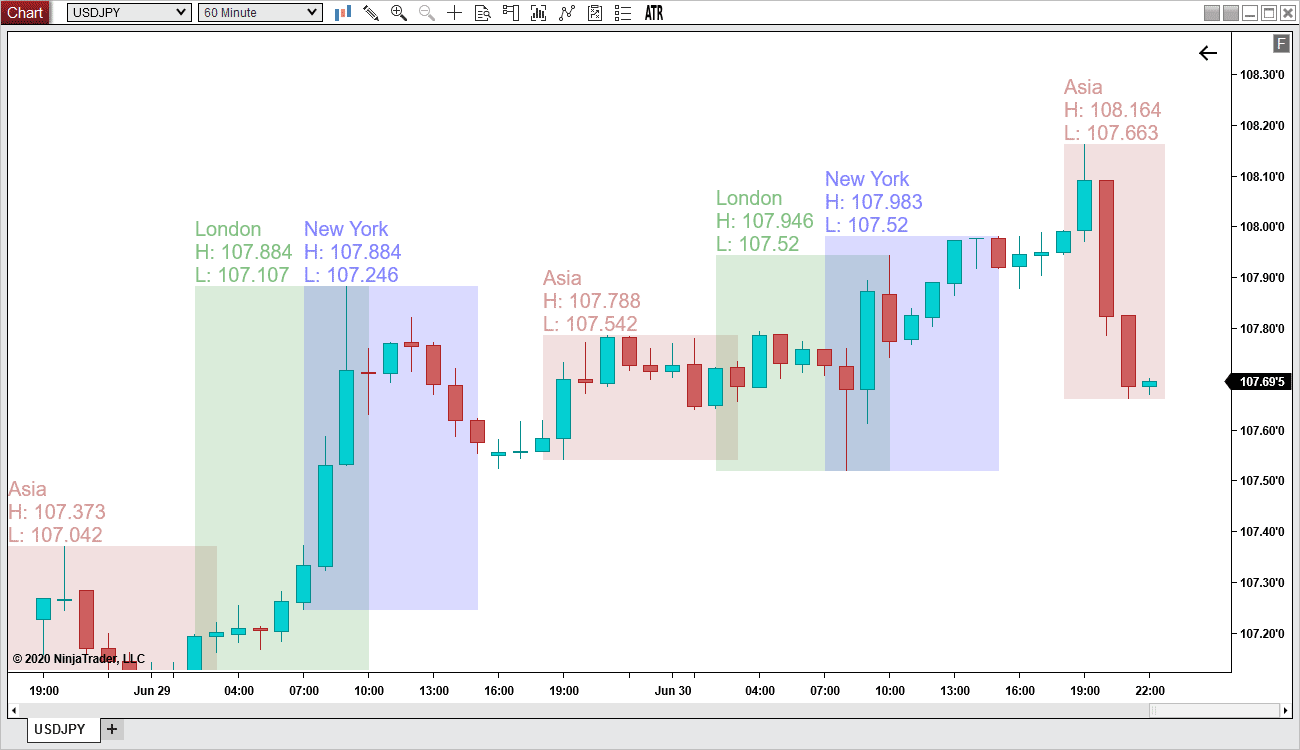
Session Hours (NT8) Chart Dynamix
Indicatormall Footprint Chart NT8 KILOCOURSE
Chart Tools (NT8) Chart Dynamix
FootPrintV2 Chart for NT8 NexusFi Forum
TDU TradeDevils NT8 Indicators Suite OrderFlow Hub
NT8 Chart PortaraNinja
Thirty Five Million Tons Of Chlorine Were Prepared By.
H _ { 2 } Gas Is Obtained At Cathode, Chlorine Gas At.
Generated At The Cathode As A Valuable Byproduct.
It Is The Technology Used To Produce Chlorine And Sodium Hydroxide (Caustic Soda), [1] Which Are Commodity Chemicals Required By Industry.
Related Post: