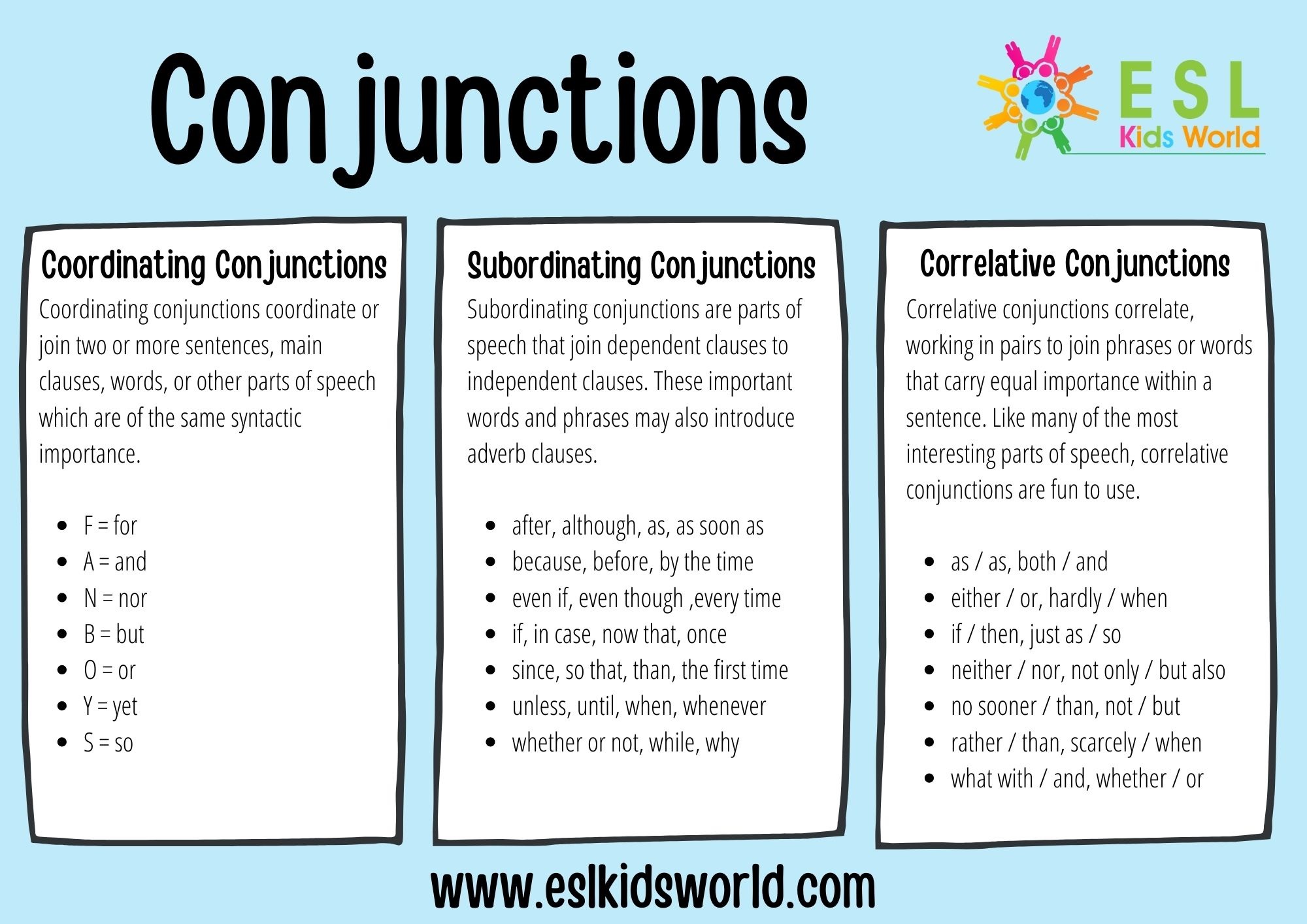
Subordinating Conjunctions Chart
Subordinating Conjunctions Chart - The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. Hence, as a result of the octet rule, the outermost energy level of an. The octet rule has significant limitations when applied to transition metals. The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. For example, elements like li, be, and b may not have eight electrons in their. These elements often form compounds with various oxidation states and can have more or fewer. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule. A stable system does not require the completion of the octet. Limitations :in the presence of one electron, hydrogen gains or loses its valence electron in order to maintain stability. However, some of noble gas. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. The octet rule has significant limitations when applied to transition metals. However, some of noble gas. The octet rule states that an atom will be most stable when its valence shell contains 8 electrons. A stable system does not require the completion of the octet. Hence, as a result of the octet rule, the outermost energy level of an. Limitations :in the presence of one electron, hydrogen gains or loses its valence electron in order to maintain stability. According to octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements. For example, elements like li, be, and b may not have eight electrons in their. The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. These elements often form compounds with various oxidation states and can have more or fewer. Limitations :in the presence of one electron, hydrogen gains or loses its valence electron. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule. The octet rule states that an atom will be most stable when its valence. These elements often form compounds with various oxidation states and can have more or fewer. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. The octet rule is adhered to by atoms because they constantly seek the most. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. The octet rule has significant limitations when applied to transition metals. These elements often. The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. The octet rule has significant limitations when applied to transition metals. Limitations :in the presence of one electron, hydrogen gains or loses its valence electron in order to maintain stability. For example, elements like li, be, and b may not have eight electrons. However, some of noble gas. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule. The octet rule states that an atom will be most stable when its valence shell contains 8 electrons. For example, elements like li, be, and b may not have eight electrons in their. The octet rule is a principle in. The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule. However, some of noble gas. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them. Hence, as a result of the octet rule, the outermost energy level of an. For example, elements like li, be, and b may not have eight electrons in their. However, some of noble gas. According to octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements. The octet rule has significant limitations when. The octet rule states that an atom will be most stable when its valence shell contains 8 electrons. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. Some atoms, especially those with fewer than four outer electrons, don’t. A stable system does not require the completion of the octet. The octet rule states that an atom will be most stable when its valence shell contains 8 electrons. However, some of noble gas. For example, elements like li, be, and b may not have eight electrons in their. The octet rule is a principle in chemistry that asserts that. The octet rule has significant limitations when applied to transition metals. The octet rule is a principle in chemistry that asserts that most chemical elements tend to react in a manner that enables them to attain a stable configuration of eight valence. Hence, as a result of the octet rule, the outermost energy level of an. These elements often form compounds with various oxidation states and can have more or fewer. The octet rule is adhered to by atoms because they constantly seek the most stable electron configuration. For example, elements like li, be, and b may not have eight electrons in their. Limitations :in the presence of one electron, hydrogen gains or loses its valence electron in order to maintain stability. The octet rule states that an atom will be most stable when its valence shell contains 8 electrons. Some atoms, especially those with fewer than four outer electrons, don’t follow the octet rule.Subordinate Conjunctions List, Subordinating Fanboys Conjunctions Examples English Grammar
Subordinating Conjunctions (Anchor Chart) in 2024 Conjunctions anchor chart, Anchor charts
Conjunctions List For Kids
Subordinating Conjunctions Anchor Chart
Subordinate Conjunctions Anchor Chart Etsy
Subordinating conjunctions RULES IN GRAMMAR Mingleish
Subordinating Conjunctions Chart
Subordinating Conjunctions Chart
Subordinate Conjunction Vs Coordinating Conjunction
Subordinating Conjunctions Chart
However, Some Of Noble Gas.
A Stable System Does Not Require The Completion Of The Octet.
According To Octet Rule, Atoms Take Part In Chemical Combination To Achieve The Configuration Of Nearest Noble Gas Elements.
Related Post: